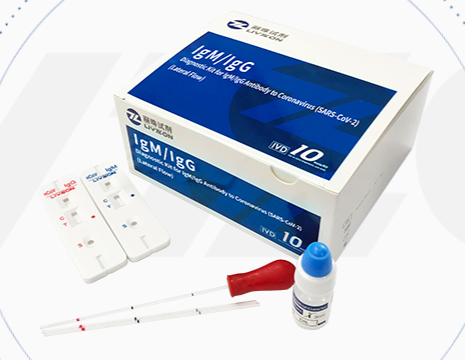
Argentina purchased 170,000 Chinese rapid test kits to monitor the spread of the novel coronavirus. A report by the COVID-19 Unit of the Argentine Ministry of Science, Technology, and Innovation concluded that the Chinese kits presented false readings, and only detect one of the two antibodies they were designed to identify.
The company Zhuhai Livzon Diagnostics, based in Guangdong, China, manufactures the test kits. The Argentine company Petroquímica Cuyo bought the kits and donated them to Argentina’s health authorities. The National Administration of Drugs, Foods, and Medical Devices (ANMAT, in Spanish) approved the kits.

With a drop of blood, the Livzon kit detects two types of antibodies: IgM (immunoglobulin M) and IgG (immunoglobulin G). Separate tests are used to find each antibody. When the result is positive, two red lines appear; when it is negative, only one line appears.
The manufacturer says on its website that it only takes 15 minutes to obtain results and that sensitivities to IgM and IgG are 90.6 percent and 99.2 percent, respectively. However, Argentina’s COVID-19 Unit concluded that the IgM detection is “weak.” “We do not recommend its use,” said the official report, according to La Nación newspaper on May 10.
Concerning IgG, Argentine scientists determined that Livzon has 80 percent sensitivity and that “50 percent of these detections showed very faint lines,” the document says. The report indicates that people must wait an additional 10 minutes to read the kit’s results in an environment with good light, and adds that “faint lines are considered positive for COVID-19,” La Nación reported. According to the newspaper, Petroquímica Cuyo paid $700,000 to buy the tests.
Only 0.6 percent of tests were positive
The Health Ministry has used the Livzon kit to monitor the spread of the virus in the train stations of Buenos Aires. Authorities aim to use rapid tests to identify asymptomatic individuals, because they estimate that two out of three people who are infected present no symptoms.
During the first stage of the epidemiological study, the kit was used on 1,270 volunteers, and delivered only eight positives (0.6 percent, according to official sources), which sparked controversy.
Specialists consulted by the local press agree that it is an extremely low number, since passengers from the Buenos Aires Metropolitan area, which has Argentina’s highest viral circulation, travel through these stations. The faulty Chinese kits might underestimate the spread of the virus among the population and compromise the flexibility of Argentina’s quarantine.
Health Ministry spokespersons told Infobae that after the kits were approved in the country, authorities decided not to use the IgM tests, due to their low sensitivity. They instructed personnel to wait 20 minutes for IgG and to consider faint lines as “positive.”
Because of these limitations, testing could not be conducted randomly at the terminals. Volunteers needed to be asymptomatic, having no fever for example, during the past 21 days.
Health Ministry sources also told Infobae that ANMAT did not approve the test for the market, but it did for this particular study. “What we are going to have is a panorama of how many people have positive antibodies in Argentina, knowing that if the sensitivity is 80 percent, there may be 20 percent more, and we also considered this data,” they said.